A secure compound is fashioned when one atom of 1 factor unites with a specific variety of atoms of one other component. All parts have totally different capabilities for mixing. Note, nonetheless, that this rule solely how many valence electrons does cr have applies to components that are not transition metals. Transition metals have more difficult electron configurations. Thus you must check out the element’s particular electron shell configuration to figure it out.
The valence electrons are within the last orbit of the atom. To decide the number of valence electrons of a particular atom, the electrons of that factor need to be organized. A valence electron is the variety of electrons current within the outermost shell or orbit of an atom.
Periodic Desk Of Parts With Valence Electrons Tendencies
We use/store this information to ensure you have proper access and that your account is safe. We could use this data to send you notifications about your account, your institutional entry, and/or other related merchandise. The JoVE video participant is compatible with HTML5 and Adobe Flash. Older browsers that don’t assist HTML5 and the H.264 video codec will still use a Flash-based video player. We advocate downloading the latest version of Flash here, however we help all versions 10 and above.
- The sharing of outer-shell electrons between atoms takes place to realize the noble gas configuration of the participating individual atoms.
- We already know what the valence electron is about.
- Let’s start by focus on ing history and discovery of phosphorus.
- However, the valence electron of the transition element can be easily determined based on the Aufbau principle.
- By determining which period the component is positioned inside determines the energy stage of the valence.
In this case, the valence electrons of copper are two. But this electron configuration in copper is not correct. The d-orbital can maintain a most of ten electrons. From the electron configuration of copper, we are in a position to see that it has 9 electrons in its d-orbital. For instance, the electron configuration of cobalt reveals that the electron configuration ends in a d-orbital and that the last shell has a total of nine electrons. Therefore, the valence electrons of cobalt are 9.
We may also learn how to tell how many valence electrons a component has. If each atom does not have an octet configuration after the lone pairs have been allocated, a double or triple bond must be drawn to meet the octet valency of each atom. Each atom in the molecule now has a lone pair of electrons assigned to it. The most electronegative atoms are often assigned the lone pairs first. In the internal shell of a transition steel, a valence electron can exist.
What’s Valence Electrons?
Valence electrons are the electrons within the highest occupied principal power degree of an atom. The last shell of an oxygen atom has six electrons. The oxygen atom completes the octave by sharing electrons with the 2 hydrogen atoms. Its outermost shell has a duplet, which is steady. The capability of an atom to combine with other atoms known as its valency.
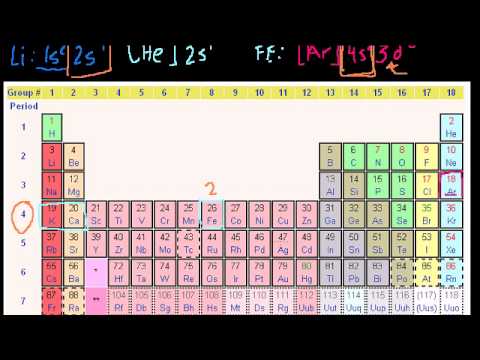
For main group elements (i.e s-block and p-block elements), the valence electrons are the electrons current within the outermost orbit. The electron configuration of argon reveals that the final shell of an argon atom has a complete of eight electrons. So, we will say that the variety of valence electrons in the component of group-18 is eight.
Actual Valued And Piece Functions: Definition, Graphs, Examples
For example, sodium, atomic quantity eleven, has 11 electrons and an electron configuration of two,eight,1. There is 1 electron in the outermost shell, so sodium has one valence electron. In this drawback, we didn’t find yourself using the second relationship between the number of electron shells and the bond energy.
